Introduction to Air Pollution
Air pollution refers specifically to the degradation of air quality within the troposphere, the lowest layer of Earth's atmosphere where weather phenomena occur and where all terrestrial life interacts with the gaseous envelope. Pollution in this layer is mainly anthropogenic and arises from diverse sources such as vehicular exhausts, industrial emissions, and agricultural practices.
The troposphere, extending from the Earth's surface up to roughly 8–15 km depending on geographical location, acts both as a reservoir and a reactive medium for pollutants. However, due to dynamic meteorological processes like wind, turbulence, and rainfall, most pollutants in this layer tend to have a short residence time. These pollutants are eventually washed out by precipitation, degraded by chemical reactions, or deposited on land or water surfaces.
What Happens when Pollutants reach directly in Stratosphere?
Although the troposphere is the principal site of pollution events, certain extreme phenomena—such as major volcanic eruptions—can inject pollutants directly into the stratosphere, the layer above the troposphere. Unlike the troposphere, the stratosphere is relatively stable and less turbulent, allowing pollutants (e.g., volcanic ash or sulfur aerosols) to linger for years.
This persistence can result in global-scale climatic effects, such as:
- Reduction in solar radiation due to atmospheric scattering and absorption
- Lowering of global surface temperatures, which is the basis of the hypothetical “nuclear winter” scenario—where widespread combustion from nuclear warfare could throw enough soot into the stratosphere to significantly block sunlight, disrupting agriculture and ecosystems.
Such events underline the difference in environmental impact and temporal scale between pollution confined to the troposphere and pollution that reaches the stratosphere.
Mechanism of Atmospheric Air Pollution
Air pollution in the troposphere is typically a result of continuous emissions from anthropogenic sources such as:
- Vehicular exhausts (carbon monoxide, nitrogen oxides, hydrocarbons)
- Industrial chimneys (sulfur dioxide, particulate matter, volatile organic compounds)
These pollutants do not remain inert. Instead, they undergo secondary reactions in the atmosphere, often in the presence of sunlight, forming more toxic compounds. A classic example includes the formation of tropospheric ozone (O₃) through photochemical reactions between nitrogen oxides (NOₓ) and volatile organic compounds (VOCs)—a phenomenon central to photochemical smog.
Thus, air pollution is a dynamic and reactive process, not merely an accumulation of emitted substances.
Dispersion and Variability of Pollutants
Pollutants do not remain static at their point of emission. Their concentration varies depending on:
- Wind speed and direction – Influences horizontal dispersion
- Thermal turbulence – Vertical mixing of pollutants
- Topography – Valleys may trap pollutants, while open plains promote dispersion
- Temperature inversions – Can trap pollutants near the surface, exacerbating pollution levels
Therefore, atmospheric pollution is heterogeneous—its severity changes with location, meteorological conditions, and time.
Defining Air Pollution: Legal and Scientific Perspectives
Merely detecting a pollutant in the air does not qualify as air pollution. The threshold for pollution is crossed only when the concentration of contaminants exceeds prescribed standards set by regulatory authorities like the Central Pollution Control Board (CPCB) in India or the United States Environmental Protection Agency (EPA). This distinction is critical in environmental science and policy because not all presence is pollution; excess is.
According to WHO (World health Organization):
“Air pollution is the contamination of the indoor or outdoor environment by any chemical, physical or biological agent that modifies the natural characteristics of the atmosphere.”
According to Air (Prevention and Control of Pollution) Act, 1981 – India
Legal Definition under Section 2(b):
“Air pollution” means the presence in the atmosphere of any air pollutant.
“Air pollutant” [Section 2(a)]:
Any solid, liquid, or gaseous substance (including noise) present in the atmosphere in such concentration as may be or tend to be injurious to human beings or other living creatures or plants or property or environment.
Key Indicators of Air Pollution
The standard set of pollutants considered indicators of air pollution includes:
- Carbon Monoxide (CO): A colorless, odorless gas released from incomplete combustion of fossil fuels, especially from vehicular exhausts. It interferes with the oxygen-carrying capacity of blood and poses serious health risks at high concentrations.
- Nitrogen Oxides (NOₓ): These include nitrogen dioxide (NO₂) and nitric oxide (NO), predominantly released from fuel combustion in vehicles and power plants. They contribute to the formation of photochemical smog, acid rain, and respiratory issues.
- Sulfur Oxides (SOₓ): Emitted mainly from the burning of sulfur-containing fossil fuels such as coal and oil, particularly in industrial processes. Sulfur dioxide (SO₂), the primary compound, can irritate the respiratory system and also lead to acid rain.
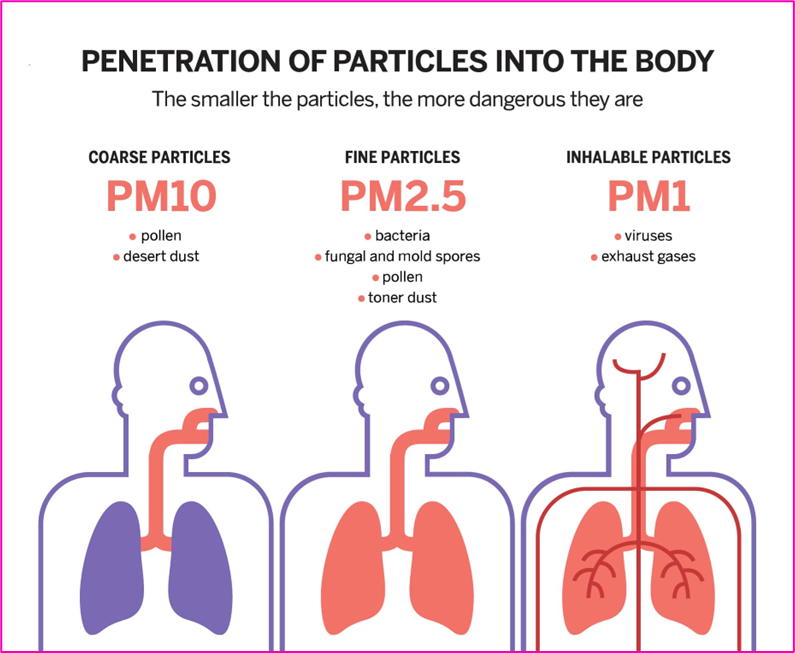
- Particulate Matter (PM): These are tiny solid or liquid particles suspended in the air. Particulates can include dust, smoke, soot, and other microscopic materials. Of particular concern is PM₁₀—particulates with an aerodynamic diameter of 10 micrometers or less—as they can penetrate deep into the lungs and cause severe respiratory and cardiovascular effects.
- Dust: Often treated as a component of particulate matter, dust includes coarse particles from roads, construction sites, and soil erosion. Though seemingly natural, in high concentrations it contributes to visibility reduction and respiratory problems.
- Ozone (O₃): Unlike the other pollutants, ozone is not directly emitted into the atmosphere. It is formed through complex photochemical reactions involving nitrogen oxides (NOₓ) and volatile organic compounds (VOCs) under the influence of sunlight. Ground-level ozone is a major component of photochemical smog and is harmful to both humans and vegetation.
Baseline Composition of Clean Dry Air
To effectively detect and measure pollution, we must know the baseline composition of unpolluted, clean air. In clean dry air, the gaseous composition (by volume) is approximately:
Gas
Percentage Volume
Nitrogen (N₂)
78.08%
Oxygen (O₂)
20.95%
Argon (Ar)
0.93%
Carbon Dioxide (CO₂)
0.03% (now ~0.04%)
In addition to these, trace gases are also present, which play vital roles in climate dynamics and atmospheric chemistry:
- Neon, Helium, Krypton, Xenon – Noble gases with inert behavior
- Methane (CH₄), Nitrous oxide (N₂O) – Greenhouse gases
- Ozone (O₃) – Beneficial in the stratosphere, harmful in the troposphere
- Carbon monoxide (CO) – Highly toxic, especially indoors
- Ammonia (NH₃) – Agricultural emissions, precursor to PM formation
- Sulfur dioxide (SO₂), Nitrogen dioxide (NO₂) – Respiratory irritants and acid rain precursors
- Hydrogen sulfide (H₂S) – Malodorous and toxic
These gases, although minute in concentration, can act as potent pollutants if emitted in large volumes or under specific environmental conditions.
Types of Air Pollutants and Their Sources
Air pollution results from the introduction of harmful substances into the Earth's troposphere due to both natural processes and human activities. These pollutants vary in form, chemical composition, and environmental behavior. Broadly, air pollutants are classified based on their source, chemical nature, and method of formation.
Air Pollutants from Human Activities (Anthropogenic Sources)
Human activities such as industrial production, vehicular transport, energy generation, and urban development have drastically increased the emission of harmful air pollutants. These are broadly grouped into two major types: particulate pollutants and gaseous pollutants, based on their physical state.
Suspended Particulate Matter (SPM)
Definition and Nature
Suspended Particulate Matter refers to tiny solid or liquid particles that are suspended in the atmosphere. These particles range in size, origin, composition, and reactivity. Depending on their size, they may remain airborne for seconds to days, travel long distances, and affect both outdoor and indoor air quality. Some particles are visible to the naked eye, such as road dust or smoke, while others are microscopic aerosols that require specialized equipment for detection.
SPM is generally classified into:
- PM₁₀ (particles ≤10 micrometers): Can penetrate the upper respiratory tract.
- PM₂.₅ (particles ≤2.5 micrometers): Can reach deep into the lungs and even the bloodstream.
SPM has both natural sources (like dust storms and sea spray) and anthropogenic sources (like vehicular emissions, industrial operations, and combustion).
Components of SPM: Detailed Description
Bio-Particles
These are biological or semi-biological entities suspended in the air, originating from both natural ecosystems and human-modified environments.
- Examples: Bacteria, viruses, fungal spores, pollen grains, insect debris.
- Sources: Agricultural fields, forests, stagnant water bodies, composting sites, and indoor ventilation systems.
- Impacts:
- Cause allergic reactions, including hay fever and asthma.
- Serve as pathogen carriers, contributing to airborne diseases like tuberculosis and influenza.
- In occupational settings (e.g., grain silos or compost plants), they can trigger hypersensitivity pneumonitis and chronic lung disease.
Dust Particles
Dust is a heterogeneous mix of mineral and organic particles, generally coarse in size, though some components fall under the PM₁₀ or PM₂.₅ category.
- Sources: Road transport, construction activities, demolit
- Causes respiratory irritation, coughing, and exacerbation of existing lung conditions like asthma and bronchitis.
- Contributes to reduced visibility and soiling of surfaces.
- Can carry adsorbed toxic substances, increasing their health risk upon inhalation.
Soot/Black Carbon
Soot, also known as Black Carbon (BC), consists of fine carbonaceous particles formed from the incomplete combustion of fossil fuels, biomass, and other organic matter.
- Examples: Diesel exhaust, smoke from biomass burning, industrial emissions.
- Sources: Vehicles (especially diesel engines), power plants, industrial boilers, residential heating (wood/coal burning), agricultural burning, and forest fires.
- Impacts:
- A significant component of PM₂.₅, leading to severe respiratory and cardiovascular diseases, including lung cancer, heart attacks, and strokes.
- A powerful short-lived climate pollutant, it absorbs solar radiation, contributing to global warming and regional climate change by warming the atmosphere and accelerating ice melt.
- Reduces visibility and causes widespread soiling of buildings and infrastructure.
Correct Answer: c) Ozone (O₃)
Explanation: Ozone in the troposphere is primarily a secondary pollutant, formed by photochemical reactions involving NOₓ and VOCs.
Correct Answer: b) Incomplete combustion of fossil fuels and biomass
Explanation: Black carbon is formed when fuels like diesel, wood, or coal are burned inefficiently, releasing fine carbon particles into the atmosphere.
स्रोत: LearnPro Editorial | Environmental Ecology | प्रकाशित: 6 October 2025 | अंतिम अपडेट: 12 March 2026
लर्नप्रो संपादकीय मानकों के बारे में
लर्नप्रो की संपादकीय सामग्री सिविल सेवा तैयारी में अनुभवी विषय विशेषज्ञों द्वारा शोधित और समीक्षित है। हमारे लेख सरकारी स्रोतों, NCERT पाठ्यपुस्तकों, मानक संदर्भ सामग्री और प्रतिष्ठित प्रकाशनों जैसे द हिंदू, इंडियन एक्सप्रेस और PIB से लिए गए हैं।
सामग्री को नवीनतम पाठ्यक्रम परिवर्तनों, परीक्षा पैटर्न और वर्तमान घटनाक्रमों के अनुसार नियमित रूप से अपडेट किया जाता है। सुधार या प्रतिक्रिया के लिए admin@learnpro.in पर संपर्क करें।